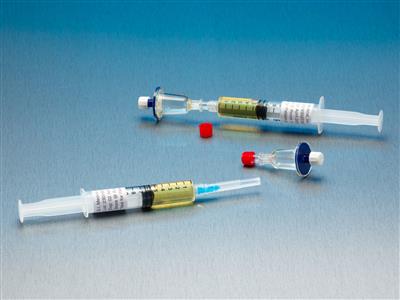
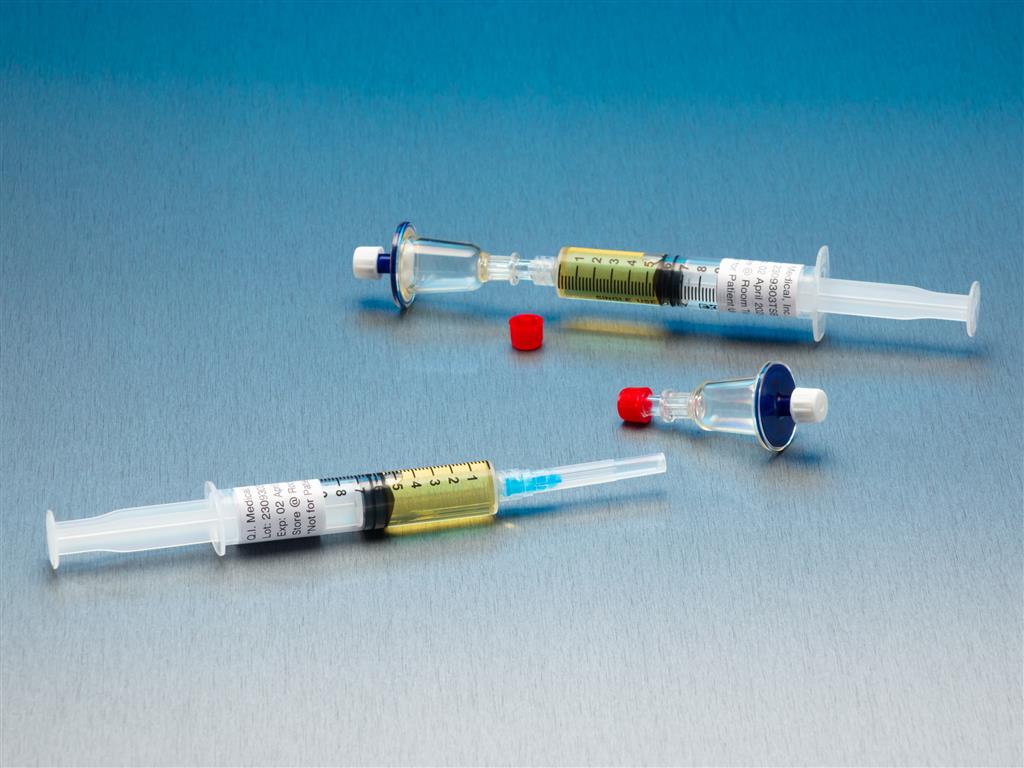
Qt Micro System, 0.22 Contamination tester, female luer lock inlet, male luer slip outlet, TSB Medi,
Item No.
TM6000
Qt Micro System Full filtration microbial contamination test system for small volume compounded sterile products. Nondestructive test allows for sample to be used after testing. FDA Registered Class 2 Device with required FDA Unique Device Identifier (UDI) barcode. 0.22µ contamination tester for
Description
Qt Micro System
Full filtration microbial contamination test system for small volume compounded sterile products.Nondestructive test allows for sample to be used after testing. FDA Registered Class 2 Device with required FDA Unique Device Identifier (UDI) barcode.
- 0.22µ contamination tester for syringe to syringe transfers.
- Log sheet included
- Gummed labels included
- 10 x QT Micro filter units with female luer lock inlet and female luer lock outlet
- 10 x 5cc syringes of TSB growth media
Reviews (0)
There are no reviews yet.
100ml EVA 2-Port Bag Non-DEHP, Latex Free
Item No.
66040
Avert™ Surface Disinfectant Cleaner Liquid 32 oz. Bottle
Item No.
DVO100842725
Tacky Mat - 18" x 36" Gray 60 Layer 4/case
Item No.
PM1836-64-G